Dirty to clean: best practice in handpiece decontamination
What measures should practices adopt to ensure their handpiece decontamination protocols meet or even exceed best practice standards?
An essential element of effective infection prevention and control in a dental practice is the adoption of effective handpiece decontamination, especially in these challenging times. The hidden internal surfaces and hollow channels (lumens) of dental handpieces present a particular challenge to the decontamination team who must be sure that they can reliably and reproducibly deliver handpieces which have been effectively cleaned both internally and externally and sterilised back to the surgery for safe re-use.
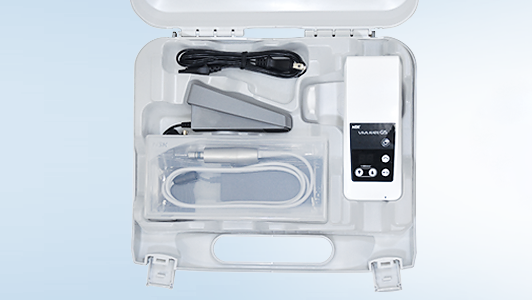
To help practices achieve this, the NSK SMART decontamination strategy has been devised, which takes the three crucial decontamination steps of handpiece cleaning, lubrication and sterilisation and helps practices to deliver these in a Safer, Measured, Affordable, Reproducible and Time-saving approach.
Following these steps removes the risks of error and inconsistencies typical of a manual approach by advocating the use of fit-for-purpose equipment which has been specifically designed to facilitate each part of the process, safely and effectively. The automated cleaning, lubrication and sterilisation processes that these pieces of equipment undertake ensure not only that the guidance is exceeded but that the desired outcomes can be reproduced every time.
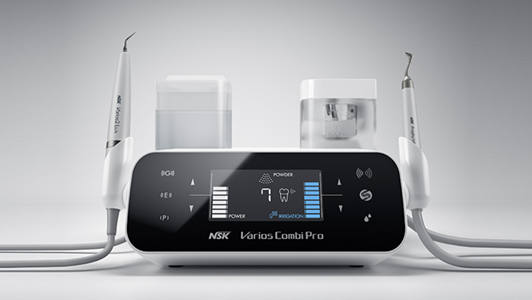
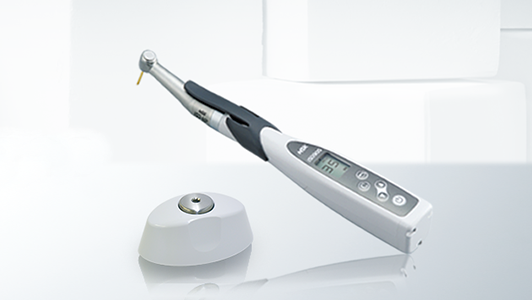
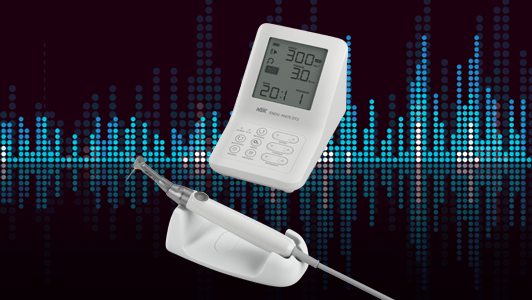
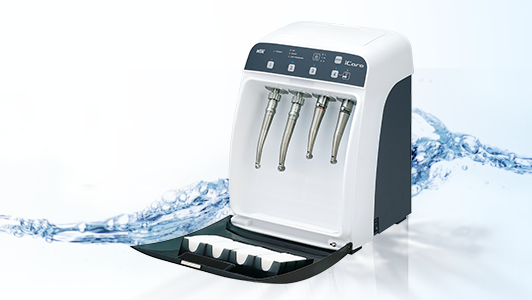
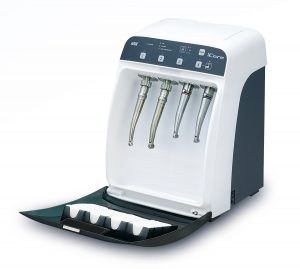 An automated instrument cleaning and lubrication unit, such as the NSK iCare, assures correct reproducible lubrication every time. It also ensures complete removal of moisture, debris and excess lubricant by the use of compressed air at every stage, which automatically purges the handpiece in a matter of seconds. This step is essential to allow complete steam penetration during subsequent sterilisation.
An automated instrument cleaning and lubrication unit, such as the NSK iCare, assures correct reproducible lubrication every time. It also ensures complete removal of moisture, debris and excess lubricant by the use of compressed air at every stage, which automatically purges the handpiece in a matter of seconds. This step is essential to allow complete steam penetration during subsequent sterilisation.
Ensuring the effective sterilisation of hollow dental handpieces is one of the most complex challenges faced by decontamination teams. An autoclave must be able to achieve full steam penetration of all surfaces and to maintain a sufficiently high temperature for both sterilisation and drying phases. Any air that remains trapped within an instrument or load will act as a barrier to steam penetration and result in a failure to sterilise.
Despite the lack of clear guidance in this area, there is evidence from Winter (2016) that clearly supports the fact that hollow instruments cannot be sterilised in a class N, non-vacuum autoclave. Non-vacuum autoclaves rely on gravity or downward displacement of air (passive air removal) which is not effective at removing all the air, especially from hollows including handpiece lumens, before steam is introduced.
Class S and B vacuum autoclaves, on the other hand, use a built-in vacuum pump which sucks out all the air from the chamber and load (active air removal) prior to introducing steam. The resultant vacuum ensures the steam can fully penetrate the chamber and load and therefore sterilise instrument surfaces, both internally and externally.
After sterilisation, the thoroughly dried handpieces can immediately be placed in sterile containers ready to be transported back to the surgery. If processed in sealed sterilising pouches, instruments can be stored for up to one year, protected from bacterial recontamination or corrosion by the fast and efficient drying cycle of these thermally efficient autoclaves.
Dental handpieces are valuable pieces of equipment without which dental professionals are unable to practise. Aside from the need to safely and reproducibly decontaminate them after each use, they must also be protected as an investment. Applying the correct decontamination principles fulfils both these requirements by allowing handpieces to be reprocessed to the highest standards as well as maintaining them in excellent working order to maximise their longevity.